A new experimental study, published in Evolution Letters, reveals that the loss of predators from communities can make it more difficult for prey species to adapt to uncertain future environments. Lead author Dr Matt Barbour tells us more.
We are in the midst of the Earth’s sixth mass extinction. As an ecologist, I find this troubling because we know that different species play different roles in maintaining healthy ecosystems — ecosystems that not only inspire us with their natural beauty, but that we also critically depend upon for the air we breathe, the water we drink, and the food we eat. At a certain point, the extinction of species and local populations can cause dramatic changes in ecosystems — often degrading their natural beauty and function and, as a result, our own physical and mental health.
I’m also an evolutionary biologist, and I became curious to know how the extinction of species from an ecosystem would affect the ability of the remaining species to evolve and adapt to their future environment. On one hand, the extinction of species may make it easier for the remaining species to evolve, which could buffer the negative effects of extinction on the ecosystem. On the other hand, being part of a diverse ecosystem may provide essential “fuel” for species to adapt and persist in an uncertain and changing world. In this scenario, the extinction of species would make it more difficult for the remaining species to evolve and adapt, putting ecosystems at even greater risk.
As a first step toward solving this puzzle, myself and others conducted a field experiment that simulated the extinction of a group of wasps who parasitize a tiny fly. This miniature ecosystem of the fly and wasps is fascinating, and I want to take a minute to explain it before I describe the experiment.
The Gall Fly Ecosystem
Fly babies are called larvae, and when larvae of this fly feed on a plant, they cause the plant to grow these tooth-shaped structures called galls. The larvae live inside these galls, where they are protected from being attacked by predators, such as ants or spiders. However, a special group of wasps called parasitoids have figured out ways to circumvent the protective shield of the gall.

Biology of the gall fly Iteomyia salicisverruca. On the left, a female fly is laying an egg inside the leaf of the willow Salix hookeriana. After hatching, the fly’s larvae feed on the plant, which causes the plant to develop a gall (center photo). Each fly larva lives in a chamber inside the gall (right photo).
For example, one type of wasp has figured out how to find the fly’s eggs before they hatch and form the gall. This wasp is called an egg parasitoid, because it lays its egg inside the fly’s egg (!!!), and the wasp’s egg waits until the fly larva is older to hatch and devour it from the inside out (ever see the movie Alien? … that’s basically what happens).

The egg parasitoid finds its host. The egg parasitoid Platygaster sp. is able to find fly eggs before they hatch and develop into a gall. The parasitoid in this photo is laying its egg inside the egg of a fly.
Another group of wasps attack the fly at the larval stage and are called larval parasitoids. They find the gall after it has already developed and use a long needle-like part of their body to pierce through the gall, stun the larva inside, and lay their egg … which eventually hatches and devours the larva. Often, larval parasitoids will parasitize a fly larva that has already been parasitized by an egg parasitoid (!!!), in which case their young eat both.

The gall fly ecosystem. Gall fly larva (center) are attacked by an egg parasitoid (left) and three different types of larval parasitoids (right). Note the long needle-like body part (its ovipositor) of the larval parasitoid in the middle (Torymus sp.). The ovipositor of the other parasitoids is hidden underneath their abdomen.
The Experiment & Our Results
In our experiment, we used fine-mesh bags to prevent the larval parasitoids from attacking the fly larva. We could not exclude the parasitoid that attacks the fly’s egg, because it’s virtually impossible for human eyes to find these tiny eggs on the plant. As a control, we allowed both kinds of parasitoids to attack other galls. We flagged these control galls with tape so we could find them again later.

Manipulating the gall fly ecosystem. We used fine-mesh bags to prevent larval parasitoids from attacking the fly (left and top right). We marked other galls that could be attacked by both types of parasitoids with a piece of tape (lower right).
At the end of the summer, we collected these galls, brought them back to the lab, and dissected them to figure out which fly larva survived and which ones were killed by a parasitoid. We also took data on each gall’s traits, such as the size of the gall, the number of larvae within a gall, and the fly’s preference to create galls on particular plants. We measured these traits because we knew from previous work that each of these traits influenced the likelihood of a fly surviving from parasitoid attack.
We found that a unique combination of traits helped fly larva survive when the larval parasitoids were removed. Fly larvae were more likely to survive if they were by themselves inside a large gall and if there weren’t many neighboring galls on the same plant. This suggests that, if the larval parasitoids went extinct, the fly would evolve to have this unique combination of traits — a unique combination which helps them survive. In other words, there is only one optimal solution for a fly’s survival when larval parasitoids are gone.
In contrast, when both types of parasitoids were present, we found that different combinations of traits equally helped flies survive. For example, it didn’t affect its survival if the fly larva was by itself or with many larvae inside the same gall. Also, larvae were more likely to survive if they had either a few or a lot neighbors (but an intermediate amount of neighbors was bad news). In other words, when both types of parasitoids were present in the fly’s ecosystem, the fly had more potential solutions (i.e. trait combinations) for survival.
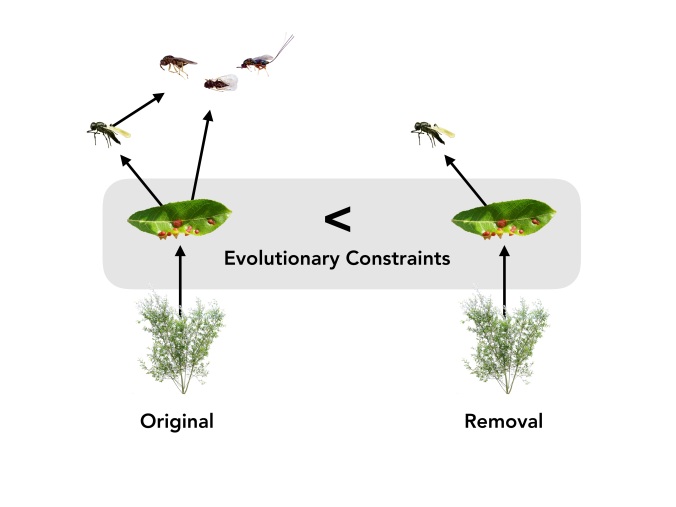
Summary of the experiment’s results. You can view the full paper here: https://doi.org/10.1002/evl3.170.
This diversity of potential solutions for survival preserves genetic variability in the traits of the fly’s galls. And since we know that genetic variation is the essential ingredient that enables a species to evolve and adapt, our findings suggest that the extinction of the fly’s parasitoids may make it more difficult for it to adapt to a changing environment.
Although we only studied a specific miniature ecosystem, we know that virtually all species on Earth have one or more “natural enemies”. Being attacked by a diverse group of natural enemies may preserve a species’ genetic diversity, which (indirectly) helps it adapt to an uncertain future. This also means that the extinction of natural enemies could make it more difficult for the remaining species to adapt and persist. If this is true, this would put many ecosystems at even greater risk than we currently realize.
Dr Matt Barbour is a Postdoctoral Research Associate at the University of Zurich. The original study is freely available to read and download from Evolution Letters.